In Sweden, the preschool curriculum states that “the preschool should strive to ensure that each child develops his/her understanding of science and relationships in nature, as well as knowledge of plants, animals, and also simple chemical processes and physical phenomena” [13, p. 10]. However, scholars like Sundberg et al. [Sundberg, 2016] have described how some preschool teachers struggle to apply the curriculum in their current preschool practice. The lack of studies focusing on chemistry-specific content within preschool settings motivated this study, which aims to tackle how abstract concepts like “atoms” and “molecules” can be introduced to preschool children.
This project is based in cultural-historical theory (CHT), which is devoted to describing “the origin and development of higher mental functions” [22, p. 83]. In order to achieve this, Vygotsky created the genetic approach with the aim to recreate the whole process of development.
From this perspective, development is regarded as a complex process of qualitative change (as opposed to organic growth or the sum of quantitative changes), and the social environment is the source of the psychological development of higher mental functions, taking place through cultural signs (like language diagrams or conventional signs) and sign mediation (i.e. knots for memory) [Veresov, 2010].
Within this framework, cognition is regarded as a collaborative process [Rogoff, 1998], and learning can be defined as a change in the child’s “relation to another person and activities in specific settings” [10, p. 183].
The interest then lies on the children’s own interpretations of science, known as emergent science, which “focuses on the development of emergent conceptions of the nature of science and the development of positive dispositions” [Siraj-Blatchford, 2001].
Cultural-historical concepts relevant for this study
First of all, with relation to the age of the participants in the study, the social situation of development needs to be considered, as it determines “the place within the system of social relationships that children of a given age occupy, the demands that society places on them in association with this, and the rights and responsibilities that are associated with this” [1, p. 84]. With this regard, since play is considered a key activity in the preschool child’s development and the “child’s activity in an imaginary situation liberates him/her from situational constraints” [27, p. 11], a play-based learning intervention has been selected.
Secondly, among the various definitions of the zone of proximal development (ZPD), the following one has been adopted; “ZPD is the field of possibilities that the child discovers in the process of collaboration with the adult” [Rubtsova, 2019]. This means understanding that the levels of actual development are not determined, but identified, with the help of tasks, and “the process of development is “triggered” only when the very process of learning, i.e., joint solution of problems, begins to be built in the zone of proximal development” [Veresov, p. 28].
For this reason, a more active role of the preschool teacher guiding the child’s play has been adopted, following the sustained shared thinking pedagogy, described as “an interaction where two or more individuals ‘work together’ in an intellectual way to solve a problem, clarify a concept, evaluate activities, or extend a narrative” [Sylva, 2004, p. 718].
Last but not least, there are the important definitions of everyday or spontaneous concepts as the ones that “emerge from the child’s everyday life experience” [Vygotsky, 1987, p.172] and scientific or non-spontaneous concepts that “can arise in the child’s head only on the foundation provided by the lower and more elementary forms of generalization which previously exist” [Vygotsky, 1987, p. 177]. The difference is crucial, because “the learning of scientific concepts depends on the concepts developed through the child’s own experience” [Vygotsky, 1987, p. 180]. For this reason, conceptual play [Fleer, 2009], which integrates scientific concepts within preschool children’s everyday practices, as everyday and scientific concepts relate to each other in a dialectical way, has guided the design of the intervention.
Besides the theoretical concepts that explain the process of origin and development of higher mental functions (like its sources, character and moving forces), the cultural-historical theory is accompanied by a set of experimental tools that form the experimental-genetic method [Veresov, 2014]. This method has as its main feature the combination of descriptive and explanatory tasks, which helps differentiate processes that can have similar external manifestations but the underlying connections do not coincide.
For this project, the principle of “buds of development” is particularly important since it focuses on experimental studies that take emergence into consideration, stating that research should begin by describing the object of study when it is in its “bud” (its early and not yet developed stage) [Veresov, 2014, p. 139]. In this study, “buds of development” are equivalent to emergent science [Siraj-Blatchford, 2001], which is seen as preceding scientific concepts, and it can be all the instances of reasoning that children express when trying to understand a science-related issue. The interest lies particularly in the way “buds of development” are changing in the process of cooperation through the dialogue with the teacher.
Category, which in Russian (“категория”) means a collision, contradiction or dramatical event [Veresov, 2014], is another principle from the genetic-research method that is useful for this study. O. Rubtsova and H. Daniels [Rubtsova, 2016] explain the origin of this term by the influence that L.S. Vygotsky’s early work as theatre reviewer had in the development of his psychological theory. This finding came from studying the new publication of his private archives, which allowed for a reconceptualization of Vygotsky’s theory that “challenges the mainstream interpretations” [Dafermos, 2018].
According to L.S. Vygotsky [Vygotsky, 1997, p. 106]:
Genetically, social relations, real relations of people, stand behind all the higher functions and their relations. From this, one of the basic principles is ...of experimental unfolding of a higher mental process into the drama that occurs among people.
These categories, which are like collisions or contradictions, have guided the selection of vignettes of this study, in which children correct themselves and realise that there are other ways to explain phenomena that puzzle them.
The principle of developmental tools, which refers to the cultural signs that the child discovers “in cooperation with an adult or more competent peer” [Veresov, 2014, p. 89], has been applied in order to analyse the role that mediating tools have played in supporting the child’s understanding of the concepts of “atoms” and “molecules”.
The goal of science teaching in early childhood is defined as examining “scientific learning in relation to how everyday situations create scientific encounters, which are emotionally charged and socially mediated in actions and activities” [Fleer, 2013, p. 2086]. Within this approach, M. Fleer [Fleer, 2015] developed a cultural-historical model for preschool science education, which has guided the design of this intervention. In this particular case, the project was presented through a story that connected with the children’s interests (kings, princesses and knights), as this theme was observed to be frequent in their free- play activity during an observation period previous to the start of the intervention.
For feasibility reasons, in the pilot study, a group of four children (two boys and two girls, all native Swedish speakers) from a preschool in a small town in the southeast of Sweden were selected. The study began in January 2018. A second group was composed of 16 children (ten boys and six girls) from a bilingual preschool (Swedish and English) in a larger city. The second stage was launched in November 2018. Data presented in this paper belong to one of the groups from the second preschool.
Ethical approval was obtained from the regional ethical board and informed consent was granted from the principal, staff, parents and children involved in the project. To ensure (to the best of our ability) that the children who did not want to participate in the activities could express their discontent, regular staff were present at all times to interpret body language.
In compliance with the GDPR regulation [EU 2016/679. General], all video-recordings were saved on external hard disks, kept in a safe box, and only viewed by the members of the research group. In order to assure confidentiality of the participants, pictures of the children have been anonymised with simple sketches and names have been changed with the use of aliases.
Visual ethnography was selected as the method for collecting data, because “it is a way of researching from the perspective of being part of an environment rather than from that of asking someone to tell you about it in spoken words.” This allows the study of learning as a collective effort [14, p. 116].
With reference to the researcher’s role when conducting a study framed within CHT, a double-ness can be observed, in the sense that the person is acting as a researcher and at the same time is having a personal relationship with the children and adults in the setting. For this reason, it is especially important [Corsaro, 2016] to reflect about the researcher’s role and “make clear the object of the researcher’s goal in order to distinguish this from the researched person’s intentions and motives”, so that reliability can be guaranteed [9, p. 207].
The sessions lasted between 15 to 30 minutes, and they took place during the morning with the pilot group (9:30—10:00) and in the afternoons for the second preschool (14:00—14:30). All of them were video-recorded, transcribed and completed with field notes after the discussion with all the team (including researchers and regular staff).
To ensure validity, holistic observations, coherent with the theoretical framework, were adopted. Reliability was gained through inter-rater agreement among the researchers and using triangulation by asking the pedagogues or parents of the children for clarification in case of doubt.
The results consist of a selection of “critical episodes” that illustrate significant behaviours or something characteristic of the specific setting; [Siraj-Blatchford, 2010] deems these as useful analytical tools. In this particular case, these were moments in which children showed understanding of simple chemical phenomena.
The statement that “human action is built through simultaneous deployment of a range of quite different kinds of semiotic resources” [8, p.1489], shows a close relation to Vygotsky’s theory, which places great importance on the use of signs and symbols, such as, language. For this reason, Ethnomethodology and Conversation Analysis (EMCA) has been adopted for representing the results, because it serves the purpose of overcoming the complexity that multimodality poses on transcribing social interactions [Mondada, 2018]. The transcription conventions adapted from [Melander Bowden, 2019] can be found in the Annex.
Vignette 1 relates to a previous lesson, in which a jar illustrates the water cycle; there are rocks inside a small jar representing the ground, blue water representing the sea, and a plastic bag hanging on top representing a cloud (see fig. 1 below).

Figure 1. Water cycle experiment
Vignette 1. [group 2 — date: 20.02.2019 — session 17 — min. 1,50—9,34]
1 Teacher: What do you think the blue thing is?
2 Gloria: Water.
3 James: It is water! And people live on the green.
4 Teacher: Exactly, right (.) The green part [is] where people live on the ground, here are the stones.
5 Luke: Why has it got this colour?
6 Teacher: You know why? What colour is water?
7 Simon: Blue!
8 James: Blue.
9 Teacher: Regular water, when I open the tap, what colour is it?
10 teacher *actually showing tap water*

11 Gloria: White.
12 James: If you take a glass, it changes the colour. It will turn blue, green or yellow.
13 Teacher: Exactly, so it is not regular water because I added some colour
to it, just to see it::
14 Teacher: If the water from the sea gets warm, what happens to it?
15 James: It will turn into gas!
♦ ♦ ♦
16 James: We pretend that this is a cloud.
17 Teacher: Right! This plastic bag we pretend it is a cloud (.) and do you see what is underneath it?
18 Teacher: Can you look closer?
19 Gloria: A water drop.
20 Teacher: Right!
21 James: How did it get here?
22 Teacher: Because I placed it on the heater, is it warm or cold?
23 everyone: Warm!
24 Teacher: What do water molecules do because of the heat?
24 teacher *places the jar on the heating and shakes her body*
25 Gloria: Gas.
26 Teacher: They move more and become gas.
After the teacher introduced the jar and told the children this represented the Earth (because the children had worked on a project about the planets earlier), she asked what they thought the blue thing was (line 1). The children reply that it is water (line 2) and that people live on the green (line 3), showing that they remember and can follow the metaphor of the jar.
Then Luke asks why the water is blue (line 5), to which the teacher replies by asking the children what is the colour of water (line 6) and they answer that it is blue (lines 7 and 8). Then the teacher reacts by opening a tap in the room and showing them tap water (line 10) and asking them what colour that is (line 9). Gloria replies white (line 11), and then James adds that it changes colour depending on which container it is placed in (line 12). This leads the teacher to explain that she put colouring into regular water just to be able to see it (line 13).
Once everyone understands the jar, the teacher begins with the explanation of the water cycle and starts by asking what happens to the water in the sea when it gets warm (line 14) (because children had worked with the states of matter in previous sessions), and James replies that it turns into gas (line 15). Then after a moment, he recognises himself that the plastic bag represents the clouds (line 16). Then the teacher asks everyone to pay attention to the “cloud” (lines 17 and 18), and finally Gloria discovers that there are tiny water drops on it (line 19).
Next, James wonders how the water drops ended up there (line 20), and the teacher explains that she placed the jar on the radiator and asks them if it is hot or cold there (line 22) to which everyone replies hot (line 23). After that, the teacher asks them what happens to water molecules when they get heated (line 24). Since they do not respond right away, she gives a physical hint and starts shaking her body (line 15), which they had done in a previous activity when they pretended to be water molecules and changed among the states of matter. Gloria remembers this and says “gas” (line 16), and the teacher rephrases her answer with a complete sentence (line 17).
This vignette has several examples of the “buds of development” principle [Veresov, 2014], since the children show emerging science skills (like noticing that water takes the colour of the container it is put into, deducing that the plastic bag pretends to be a cloud, and wondering how the drop got onto the plastic bag), which will be basic for future scientific work.
Vignette 2 also belongs to a later group of activities consisting of various experiments aimed at promoting hypothesising skills. The session started by dissolving sugar in water (which they had already done in stage 1), but this time the children have a clear idea of what happens with the sugar, as shown in Simon’s explanation in line 7.
Vignette 2. [group 2 — date: 03.05.2019 — session 20 — min. 7,09—12,33]
1 Simon: The sugar went down the water!
2 Teacher: Exactly, that is what I wanted to ask you (.) Now the sugar is only at the bottom, right?
3 sue *stirring the sugar with a spoon*
4 Teacher: Look, what is Sue doing with the spoon?
Stir it, what happens with the sugar?
5 Gloria: It mixes!
6 Teacher: It mixes, ok:: 7 Teacher: What happens to the sugar then, when you mix it with water?
8 Simon: First it mixes, then it becomes tiny small
and then we cannot see it.
9 Teacher: Right!
10 Teacher: Do you know what this is?
11 teacher *showing a glucose 3D molecule model*
12 Simon: A water molecule that moves.
13 Teacher: Almost, but do they look alike?
14 teacher *places glucose molecule next to a water molecule*
15 Simon: No::
16 Teacher: Which one is bigger?
17 James: This one.
18 james *pointing at the glucose molecule*

19 Simon: And this one is small.
20 simon: *pointing at the water molecule*
21 Teacher: Exactly.
22 James: Why are there sugar molecules?
23 Teacher: Because there are atoms and molecules everywhere (.) in the water, in the sugar, in the clothes, in our body::
24 James: And in the heart?
25 james *pointing at his heart*
26 Simon: And in the arm?
27 simon *pointing at his arm*
28 Teacher: Exactly, everywhere.
Since the children were already familiar with water molecules, and questions about other things like water and food had showed up during meal time in the preschool (information provided by the preschool teach
ers), the teacher showed them a glucose molecule (line 10) and asked the children if they knew what it was (line 9). Then, Simon replied it was a water molecule that moves (line 11), to which the teacher responds by asking them if the two molecule models look alike (line 12), and they immediately recognise the differences (line 13) and compare their sizes (lines 14-20).
When the teacher tells them that it is a sugar molecule, James wonders why there are sugar molecules (line 21), and the teacher answers by explaining that we can find atoms everywhere (line 22). James and Simon seem puzzled by the discovery that we have atoms in our body, and they ask if we have atoms in our heart (line 23) and in our arms (line 25), and the teacher replies by reassuring them that there are atoms and molecules everywhere (line 27).
In this sequence, both “buds of development” and category principles [Veresov, 2014] can be observed; the first one is the children observing, comparing and classifying the size of a water and a glucose molecule (which can be considered emergent science skills), and the second one, towards the end of the vignette, is the children wondering about the existence of glucose molecules and being surprised to find out that there are atoms everywhere.
Vignette 3
In a similar way to the previous vignette, this one illustrates another experiment in which children mixed water and milk because they wanted to know what milk molecules look like. The funny thing is that even before starting the activity, as soon as he saw the water jar and the milk carton, James already explained all by himself that there are water and milk molecules and they would mix (line 1). In this same line appears a cultural reference, spooky water, which is a common way that Swedish children call milk diluted in water, because they drink both with their meals and they frequently get mixed.
Vignette 3. [group 2 — date: 06.05.2019 — session 21 — min. 1,45—10,47]
1 James: You know what? Water has water mol
ecules and milk, milk molecules (.) If you pour it in here then there are both water and milk molecules, and we call it spooky water.
2 Teacher: Exactly!
3 Teacher: How does spooky water look?
4 James: A little white and a little foggy.
5 Teacher: Exactly.
6 Teacher: What does milk look like; ice, water or gas?
7 teacher *showing at a picture of each state of matter*

8 James: £Like ice.£
9 Simon: £Yes, ice!£
10 Teacher: Does milk look like ice?
11 Simon: NO!
12 Teacher: What does it look like?
13 Sue: Water.
14 Teacher: Ice is hard, but can we knock on milk? No, it moves like water.
15 Teacher: What we talk about, water and milk molecules, are not real, we pretend, because a lot of water molecules fit in a water drop, so they are tiny. You can only see them through an atomic microscope, and a lot of milk molecules fit in a milk drop. They are tiny, too. You need to think that they are so tiny that we can drink them.
16 James: You think it is big, but it is just pretending.
17 Teacher: Exactly!
18 James: When you drink then you cannot see it, but when you pretend then you can see it.
19 Teacher: Right!
20 James: When you drink you only see white, because the glass is white, but here we have black and red.
Building on the children’s mention of spooky water, the teacher asks them if it looks like ice, water or gas in line 6. Then James jokes and responds kiddingly that it looks like ice (line 8) and when the teacher asks again (line 10), Simon exclaims that it doesn’t (line 11) and finally Sue says it looks like water (line 13).
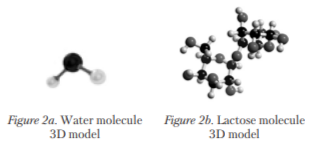
Towards the end of the session, the teacher explains that molecules are so tiny that many of them fit into a drop, so with the models they have built they just pretend these are molecules (line 15). After that, James makes three statements in his own words in which he rephrases what the teacher has suggested, explaining very well what the water and milk molecule models represent and how they are in reality (lines 16, 18 and 20) (see fig. 2a and 2b below).
This last part of the vignette is that which has motivated the selection of this session, because the explanation of how the water and lactose molecule models differ in size and colour from the real ones is an amazing example of the category principle [Veresov, 2014], in which the child has felt compelled to share his own understanding with the rest of the group very enthusiastically.
Despite the increasing interest that science in early childhood education has gained in the last decades, very
little is known about how to present chemistry content to young children. Through this cultural-historical study of a play-based learning intervention that brought chemistry to two Swedish preschools, this paper has aimed to tackle how abstract concepts like “atoms” and “molecules” can be introduced to preschool children.
Taking into consideration the social situation of development of the children, in this case, the idea of connecting with the children’s interest for kings, princesses and knights shown during their free-play, proved to be useful in introducing a chemistry play-based learning intervention, which gradually turned into more scientific experiments. In this way, the imagination that children used when creating their own fantasy worlds was directed towards understanding new everyday situations, such as figuring out that sugar is still in the water once it dissolves and we cannot see it.
Initial tasks, like the one represented in vignette 1, showed that the children could already transcend what they see and attribute new properties to materials, such as, in the Earth water cycle experiment, where a plastic bag represented a cloud, the blue liquid represented the sea water and the rocks the ground. This evidenced their actual level of development (what children were able to do by themselves).
However, the task of discussing about atoms and molecules posed too high of a challenge to the children, who had no previous experience with these concepts and that is why the use of mediating tools, like the video-animations and 3D molecular models helped them bridge the gap between what they can imagine and the reality we are talking about. Vignette 2 illustrate how the children within a joint discussion could transform the “3D molecular model of water and lactose” into “atoms we can find anywhere; like in our heart or arm”.
The proof that the intervention took place within the zone of proximal development (ZPD) is that children were able to reflect on the content discussed and formulate questions of their own, such as, “You think it is big, but it is just pretending.” (vignette 7).
Results show how the preschool children's chemistry emerges through discussion and moved from the concrete task at hand to a more general view in which atoms and molecules were extrapolated to other contexts. This was made possible due to the design of an intervention respectful of the children’s social situation of development, that integrated the learning goals into a play-based learning methodology coherent with the preschool practice children are used to. The design took the children’s actual level of development into consideration, as the children were able to solve the tasks by themselves. The mediating tools used were indeed suitable for helping the children to achieve their potential level of development [45].
Vidal Carulla C., Adbo K. Using Cultural-Historical Theory to Design and Assess a Chemistry Play-Based Learning Intervention. Kul'turno-istoricheskaya psikhologiya = Cultural-historical psychology, 2019. Vol. 15, no. 4, pp. 35—43. (In Russ., abstr. in Engl.). doi: 10.17759/chp.2019150404
Для цитаты:
Видал Карулла К., Адбо К. Культурно-исторический подход к проектированию и оценке результатов игрового обучения химии дошкольников // Культурно-историческая психология. 2019. Т. 15. № 4. С. 35—43. doi:10.17759/chp. 2019150404
[†] Clara Vidal Carulla, MD in Psychology, Natural Science Didactics PhD student, Department of Biology and Environmental Sciences, Linnaeus University, Kalmar, Sweden. E-mail: clara.vidalcarulla@lnu.se
[‡] Karina Adbo, PhD in Chemistry, Natural Science Didactics lecturer, Department of Biology and Environmental Sciences, Linnaeus University, Kalmar, Sweden. E-mail: karina.adbo@lnu.se
Клара Видал Карулла, MD (психология), аспирантка факультета биологии и экологии, Университет Линнеус, Кальмар, Швеция. E-mail: clara.vidalcarulla@lnu.se
Карина Адбо, PhD (химия), преподаватель факультета биологии и экологии, Университет Линнеус, Кальмар, Швеция. E-mail: karina.adbo@lnu.se