Spina Bifida Myelomeningocele: The Brain and Neuropsychological Outcomes
Spina bifida is the most common congenital birth defect
affecting the central nervous system in the world. One of two primary types of
neural tube defects, spina bifida has
a prevalence of about 0.5 per 1 000 live births worldwide [Copp, 2015]. The other major
neural tube defect, anencephaly (absence of forebrain), occurs with comparable
prevalence, but is lethal. Spina bifida can be identified at birth because of
the spinal malformation that gives the disorder its name ("split spine").
However, spina bifida can also be identified during gestation through
ultrasound and testing of alphafeto protein levels.
There are different subtypes of spinal defects associated with spina bifida and it is important to identify the type at birth. Myelomeningocele, accounting for about 90% of all cases, represents an open wound with the spinal cord and meninges protruding anywhere along the spinal column [Detrait, 2005]. The child sustains paraplegia below the level of the spinal lesion and varying degrees of ambulatory and urological difficulties. Other forms of spina bifida, such as meningocele and spinal lipoma (fatty tumor), are "closed" defects and primarily affect lower limb coordination and urological function, but not the brain. Although genetic factors are estimated at 60-70%, inheritance is polygenic. The most prominent environmental factor is related to deficient folate metabolism, but epidemiological research has also shown associations with maternal obesity, glucose metabolism, and exposure to pollutants and other toxins [Copp, 2015].
Myelomeningocele is the most severe form of spina bifida and is usually associated with multiple malformations of the brain. These malformations often lead to obstructive hydrocephalus that requires diversion of cerebrospinal fluid through a shunt or other approach. Myelomeningocele is often associated with cognitive and motor deficits that reflect the impact of congenital brain malformations that can be more severe because of hydrocephalus and its treatment. The malformations result from exposure of the fetus to amniotic fluid that leaks from the spinal lesion, leading to mechanical compression of other brain structures.
Brain Malformations in Spina Bifida Myelomeningocele (SBM)
A failure of neural tube closure in the first 30 days of life, SBM is a prolonged disturbance of neural migration that results in cerebellar and midbrain anomalies, abnormalities of the corpus callosum, and less frequently heterotopias and other evidence of anomalous neural migration. Figure shows an example of the brain of a child with SBM with a Chiari II malformation, beaked tectum, and hypoplasia of the corpus callosum.
The Chiari II malformation of the cerebellum and hindbrain
are virtually ubiquitous in SBM. In addition to a malformed cerebellum and
small posterior fossa, there is downward herniation of the cerebellum into the
foramen of Monro. The mechanical effects of the malformation lead to midbrain
anomalies involving the midbrain, with a tectum that is often “beaked” in
appearance and kinking of the medulla [Barkovich, 2012]. The Chiari II malformation often
obstructs the flow of CSF, leading to ventricular dilation and stretching of
axonal fibers, especially in posterior cortex and the corpus callosum. However,
in addition to
a thinned corpus callosum (hypoplasia), about 50% of children are born with
partial dysgenesis of the corpus callosum (hypogenesis) where the posterior
aspects of the body and the splenium are missing, sometimes including the
latest developing part, the rostrum. Because the corpus callosum develops
between 8 and 20 weeks of gestation, these corpus callosum abnormalities
indicate that the disruption of neural development extends beyond neural tube
closure well into later parts of gestation [Barkovich, 1988]. Hydrocephalus is often apparent
before birth on ultrasound, representing a profound disruption of early CNS
development.
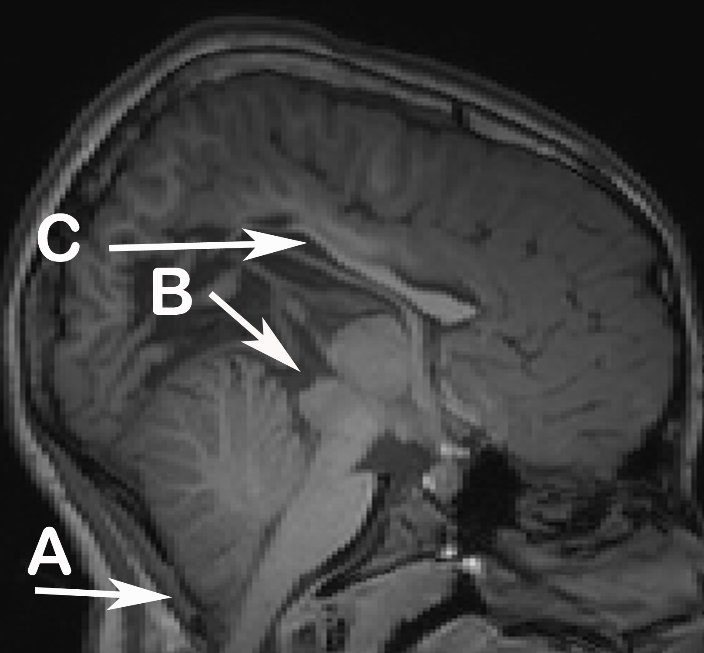
Figure. T1-weighted MRI acquired from child with SBM
Notes. A: Chiari II malformation B: Tectal beaking C: Hypoplastic corpus callosum.
Quantitative studies of the child and adult brain using structural MRI scans document atypical organization of the cerebellum and the brain [Juranek, 2010a]. On average, volumes of grey and white matter are reduced, especially in posterior brain regions, but not the frontal lobes. Here grey matter volumes are larger relative to normative values [Juranek, 2008]. The frontal, superior parietal and occipital regions are thicker, while inferior parietal and temporal regions are thinner [Juranek, 2008; Treble, 2012]. Altogether, SBM is associated with "fatter" frontal lobes and thinner posterior regions. Gyral development in SBM also shows regional differences, with higher gyrification (increased cortical folding) in the lateral frontal, inferior parietal, and posterior temporal regions and lower gyrification in the inferior frontal lobe and the medial surface of the parietal and temporal lobes [Treble, 2012].
Cerebellar volumes are lower in SBM than controls and directly related to lesion level: upper level spinal lesions thoracic and above) are associated with greater reductions than lower level lesions [Fletcher, 2005]. The atypical organization of the cerebellum is a qualitative change. The posterior-inferior regions are smaller, the anterior cerebellum is larger, and there is no difference in the corpus medullare [Juranek, 2010].
The basal ganglia and related subcortical structures are visibly normal on radiological review in SBM [Ware, 2014]. On quantitative macrostructural assessment, the hippocampus, but not the amygdala, is reduced in volume [Treble-Barna, 2015; Ware, 2014]. The putamen is enlarged. Diffusion tensor imaging of white matter structures shows that the integrity of the long association fiber tracts connecting posterior and anterior brain regions consistently reduced relative to controls [Hasan, 2008; Ou, 2011]. Reduced integrity has also been shown in the genu of the corpus callosum, but not in the anterior commissure [Herweh, 2009]. Using the midbrain as a seed point in an analysis of tectal beaking, Williams et al. found that posterior pathways showed more reduced white matter integrity than frontal pathways, especially in association with tectal beaking [Kulesz, 2015].
These studies show that the cerebrum of people with SBM is atypically organized on an anterior to posterior gradient. The mechanical effects of hydrocephalus contribute to these patterns of atypical organization. Hydrocephalus represents ventricular expansion that leads to destruction of periventricular white matter [Del Bigio, 2010]. This expansion also affects the regional distribution of white and gray matter, with cortical thickness lower along the lateral and third ventricles. However, the fact that the corpus callosum is hypogenetic in about half of children with SBM cannot be explained by hydrocephalus because the ends of the corpus callosum have simply not developed [Barkovich, 2012]. A major question involves the consequences of this atypical organization for neuropsychological functions.
Neuropsychological Consequences of Atypical Brain organization in SBM
Neuropsychological outcomes in SBM are variable. However, these variations are principled and can be understood as the outcomes of the cerebellar, midbrain, and corpus callosum anomalies, the effects of hydrocephalus and its treatment, and the consequences of the spinal lesion for movement [Dennis, 2006]. Many children with SBM show a characteristic pattern of cognitive strengths and weaknesses. Although this pattern has historically been characterized across different outcome domains, they are better understood as patterns that occur within domains because of the influence of the congenital anomalies. Thus, children with SBM have been described as stronger in verbal than nonverbal skills; better in reading than math; and as lethargic but with good social skills.
It is important to understand the pattern of cognitive strengths and weaknesses in SBM in terms of domain-specific factors within cognitive domains that are the result of domain general cognitive factors associated with the disruption of CNS development. Table shows the domain-specific factors as a modal pattern of relative strengths and weaknesses in motor, perceptual, language, reading, math, and behavioral domains. The strengths reflect the capacity for assembled processing, which we have defined as the capacity to organize, retrieve, and generate information from memory that has been associatively learned. In contrast, they have difficulties in assembled processing, which we define as constructing and integrating responses across different domains [Dennis, 2010; Dennis, 2006]. Historically, children with SBM have been reported to show preservation of syntax and vocabulary, but with poor language comprehension and pragmatics. In the framework of Table, learning vocabulary and syntax are examples of associative processing because the meaning or order of words is stipulated. Comprehension and the use of language in context requires assembling of information across multiple sources to understand or construct a response. They show better word reading and math fact retrieval, but poorer reading comprehension and math skills, again representing contrasts in which there are rules stipulating the relation (associative processing), but difficulties in content domains involving comprehension and problem-solving where information has to be assembled to construct a response. On any given task, the extent to which a child or adult with SBM will show relative strengths and weaknesses depends of the extent to which the task requires associative versus assembled processing.
Table
Strengths (associative processing) and weaknesses (assembled processing) within cognitive domains for Spina Bifida Myelomeningocele with hydrocephalus
|
|
Associative processing |
Assembled processing |
|
Domain |
Strengths |
Weaknesses |
|
Motor |
Motor Learning and Adaptation |
Motor Control and |
|
Perception |
Categorical |
Coordinate (Spatial Relations, Mental Rotations |
|
Language |
Vocabulary, Grammar |
Making Inferences, |
|
Reading |
Word Decoding |
Reading Comprehension |
|
Math |
Learning and Retrieving Facts |
Math Algorithms |
Underlying this pattern of strengths and weakness are domain general deficits in timing, attention, and motor control. These core deficits can be detected in the first year of life, persist throughout development, and have pervasive influence on cognitive and motor skills. They are directly due to the congenital malformations of the cerebellum, midbrain, and corpus callosum. Hydrocephalus can make these deficits more severe as can poverty and the treatment of hydrocephalus through the need for shunt revisions and a lifetime of medical monitoring [Bowman, 2010].
Timing and the Cerebellum
The cerebellum involves more than fine motor skills and coordination. Rather, it has a major role in role in the precision of fine motor movements that extends beyond reaching, grasping, and moving the fingers. Central to the role of the cerebellum in cognition is its role in harmonizing motor control, including rhythmicity and timing. Children and adults with spina bifida show impairment on motor free tasks involving perceptual and motor timing, and rhythm discrimination and production [Dennis, 2010a]. Cerebellar volumes are correlated with performance on these tasks. On motor learning tasks, children with SBM are impaired in performance, but not in learning. Over trials, they benefit from repetition (procedural learning), but their performance is slower and poorer in quality [Edelstein, 2004]. Thus, motor learning, which is mediated by the basal ganglia, is relatively intact, but performance and automaticity, which are more related to the cerebellum, are impaired because of the cerebellar malformation that is part of the Chiari II malformation [Dennis, 2010a]. In a randomized trial of the long-term effects of prenatal surgery for myelomeningocele, which often prevents or alleviates the Chiari II malformation, children who underwent prenatal surgery showed better (but not intact) performance than children who had postnatal spinal repair on a task requiring precision of fine motor movements [Houtrow, 2020]. They were also less likely to develop hydrocephalus requiring CSF diversion. Although the prenatal and postnatal surgery groups showed similar cognitive profiles reflecting the patterns in Table, secondary analyses need to target outcomes in those children in the prenatal group that did not develop a Chiari malformation and need a shunt.
Involuntary Attention and the Midbrain
Attention represents a dual system in the brain that involves voluntary and involuntary processes. The voluntary processes are top down and involve response control, a regulatory type of attention. Involuntary attention is stimulus driven, representing arousal, disengaging, and shifting attention as the environment changes [Dennis, 2008]. The former attention network represents an anterior system involving frontal lobe regions with connectivity into the parietal regions, while the latter is more posterior and involves the midbrain and the posterior parietal region. Children with spina bifida have abnormalities of the midbrain as a consequence of the brain developing in a smaller posterior fossa related the Chiari II malformation. Well-documented deficits in disengaging and shifting involuntary attention in SBM are associated with tectal beaking and posterior brain volume loss. Using DTI, Williams et al. those with SBM exhibited reduced tectal volumes [Williams, 2013]. There was decreased integrity of posterior but not anterior tectocortical white matter pathways, with more integrity of frontal white matter pathways. Tectal beaking was associated with lower volumes and less integrity of anterior and posterior tectocortical pathways. In an assessment of the relation of these DTI findings and measures of involuntary attention, Kulesz et al. found significant associations of slower stimulus orienting with reduced tectal volume; slower conflict resolution with reduced superior parietal cortical volume; and reduced attentional control with reduced along both frontal and parietal tectocortical pathways [Kulesz, 2015]. In contrast, Brewer et al. [Brewer, 2001] and Swarthout et al. [Swarthout, 2010] found that will children with SBM do not perform like typically developing children on measures of sustain attention (continuous performance tasks classically used as measures of response control. However, their errors occur earlier in the task and are not as significant as the difficulties on measures of stimulus control.
Movement
Children with SBM have multiple difficulties with movement related to the consequences of the spinal lesion and the cerebellar malformation. These problems are related to the core deficits in timing, but affect multiple systems from early in development. A child with SBM is born with reduced movement capabilities. They are less able to explore their environment and more likely to point and imitate than to reach and grasp [Morrow, 1992]. Dennis and Barnes [Dennis, 2010] identified multiple areas in which movement is impaired: eye movements and saccadic adaptation; visual fixation; adaptive ballistic movements of the arm in relation to vision; a variety of adaptive motor learning tasks; pacing of speech and fluency; and others involving the eyes, hands, arms, and the speech apparatus. The core deficits in movement are closely related to the timing and attention core deficits. Attention always requires movement and the quality of movement depends on timing and rhythmicity. Taylor et al. demonstrated that infants with SBM were impaired in their ability to reach and manipulated a mobile of the sort that would hang over a crib [Taylor, 2013]. The infant must fixate on it, attend to it, disengage attention in response to touching it, and adapt motor movements of the upper limbs in relation to changes in position, shape, and color. Children with SBM have difficulties with all these functions.
Interhemispheric Integration and the Corpus Callosum
Contributing to the core deficits in timing, attention, and movement and the effects on assembled processing are deficits in interhemispheric integration that stem from hypogenesis and severe hypoplasia of the posterior corpus callosum. Children with hypogenesis or severe hypoplasia of the posterior corpus callosum show reduced interhemispheric transfer on tactile tasks that stimulate either the right or left hands and on dichotic listening tasks where syllable are presented simultaneously to both ears. Performance on the dichotic listening task was correlated with total splenial area on the midsagittal measurement of the area of the splenium [Hannay, 2009]. In a study of the comprehension of metaphors, children with severe hypoplasia or hypogenesis had more difficulty understanding metaphorical language that did not have a literal meaning [Dennis, 2010]. Bradley et al. performed diffusion tensor imaging (DTI) of the corpus callosum on a large sample of children and adults with SBM and age-matched controls [Bradley, 2016]. She found reduced macrostructural and microstructural integrity of the corpus callosum in people with SBM. Interhemispheric connections were rerouted through the anterior commissure, with reduced connectivity of the posterior regions in the right and left hemisphere that was related to interhemispheric transfer on thee dichotic listening task.
Subcortical Structures, Prospective Memory, and Cognitive Control
Prospective memory, the ability to remember future intentions, is a metacognitive skill that involves remembering to remember. In contrast, episodic memory is the retrospective memory for specific events and learned material. Treble-Barna et al. found poorer prospective and episodic memory skills across the lifespan in children and adults with SBM [Treble-Barna, 2015; Treble-Barna, 2014]. Memory skills were associated with lower hippocampal (but not amygdala) volumes in children and adults with SBM. Ware et al. found that adults with SBM had significantly poorer performance on measures of working memory and inhibitory control than controls [Ware]. Those with SBM had enlarged cortical and putamen and reduced hippocampus volumes. Volumes of the dorsolateral prefrontal and hippocampus inversely correlated with performance on measures of cognitive control.
Consequences of Hydrocephalus
Hydrocephalus is a problem in SBM as a potentially lethal secondary consequence of hydrocephalus that may require shunting or another diversionary procedure, but always requires monitoring [Bowman, 2010]. In two studies, Hampton et al. compared cognitive performance on verbal and nonverbal cognitive and motor skills in groups with (a) SBM; (b) a group largely composed of children with meningocele, spinal lipomas, and other forms of spina bifida with no Chiari malformation or shunts; (c) children largely myelomeningocele and arrested hydrocephalus that did not require shunting; and (d) a group with a different etiology of congenital hydrocephalus (aqueductal stenosis) [Hampton, 2013; Hampton, 2011]. Children with aqueductal stenosis performed highest across tasks, while children with SBM had the lowest performance. Those with arrested hydrocephalus performed more similarly to the group with SBM, while those with unshunted hydrocephalus performed more similarly to those with aqueductal stenosis. However, the profiles in the groups with SBM, aqueductal stenosis, and arrested hydrocephalus were essentially parallel, suggesting that hydrocephalus expresses itself in increased severity of the patterns of strengths and weaknesses across these groups. The exception was in the motor domain, where children with SBM had more difficulties with fine motor control, likely because of the Chiari II malformation.
Clinical Outcomes
The modal cognitive profile does not occur in every child or adult with SBM. It is an average and a relative pattern where deficits on tasks involving assembled processing are more severe than on tasks involving associative processing. If there is poverty, severe hydrocephalus, problems with shunt treatments or the Chiari malformation, or other neural insults, the strengths in associative processing are less likely to appear. In terms of commonly recognized clinical outcomes, intellectual disabilities occur primarily in children who are economically disadvantaged with upper level spinal lesions [Fletcher, 2005]. In the United States, where the prevalence of SBM is high in Latinx populations who also tend to be poor, the incidence is much higher in those with thoracic level spinal lesions, reflecting the double dose of an upper level spinal defect and more severe brain involvement and poverty. Behaviorally, children with SBM are interested in people and have low levels of low levels of social anxiety. However, they can be hypersocial, overly talkative, and sometimes are seen as disinhibited because of their social and verbal behavior. In contrast, in nonsocial situations, they can appear lethargic and underaroused, which is often misidentified as amotivation. As children with SBM into adolescence, social adjustment difficulties emerge because of school, family, and peer issues [Holmbeck, 2006]. Early in school, children with SBM are often seen as very capable because of their preserved language skills and word decoding abilities. However, learning and attention difficulties are common. Burmeister et al. reported that one-third of children with SBM had elevations on parental ADHD ratings of inattention, but not hyperactivity-impulsivity. In the same cohort, 3% had word decoding problems; 26% had math problems; and 23% had both math and reading difficulties [Burmeister, 2005]. Comprehension problems were higher [Fletcher, 2005]. Adults with SBM are under-employed relative to their IQ and literacy levels. Even accounting for orthopedic impairments, Hetherington et al. found that functional math (keeping a checkbook) and spatial skills (reading maps in relation to bus schedules) were the best predictors of independent living [Hetherington, 2006].
Conclusions and Implications for Clinicians
It is important to understand and evaluate people with SBM from the perspective introduced by the modal cognitive profile. It should be expected that a person with SBM will show strengths in associative processes and weaknesses assembled processing within cognitive domains. If the profile is different, a clinician should try to understand the basis for deviations in the modal cognitive profile. Information that can be learned by repetition, memorization, repeated exposure, and other examples of associative earning can be strengths in children and adults SBM. In contrast, if the task requires integration and construction of material that is read or heard, more assistance will be needed.
People with spina bifida may require special programming in school, along with rehabilitation of their orthopedic problems. They typically require the services of an urologist. Neurosurgical intervention and follow-up begins at birth. Psychological treatment becomes increasingly important as the child moves into adolescence, but parents need education and assistance as early as possible. Comprehensive psychological assessments should begin early in development as part of a multi-disciplinary perspective [Fletcher, 2008].
Adaptive behavior assessments of functional, habitual everyday skills in conceptual, social, and daily living areas are very helpful. These are accomplished through interviews and checklists. Motor functions should be assessed in terms of their consequences for overall adaptation. Attention problems are common and more likely to reflect difficulties with inattention and arousal, although the child with SBM may have trouble accessing cognitive control processes. Assessments of academic skills are needed because many children with SBM need help with math, writing, and reading and listening comprehension.
In programming for the child, few interventions are specific to people with SBM. Interventions that address academic problems, adaptive behavior, and psychosocial difficulties in children with developmental learning and attention disorders are useful so long as the clinician understands the research base on spina bifida. It is important to build on strengths, such as those in associative processing. Focusing on generalization across contexts will assist with associative processing weaknesses. Children with SBM less likely to respond to stimulant medication if they meet criteria for ADHD [Davidovitch, 1999]. However, helping the parents and the school organize the environment and develop consistent routines. Perhaps most important, those working with SBM need to help others understand that the child has a neurological disorder that causes learning and attention problems. Many children who struggle in school or in the community are seen as lazy or amotivated, which is often not the case and rarely a satisfactory answer as to why the child or adult with SBM is struggling. These factors exacerbate learning and adjustment, but they are secondary to the brain-based difficulties with learning.