INTRODUCTION
Bipolar disorder (BD) is a mental disorder that causes impairments in the functionality of daily life, resulting in substantial burdens upon affected individuals, their caregivers, and society at large [1, 2]. Despite the therapeutic advances achieved to date, BD remains one of the mental disorders with the severest burden around the world [3]. People with BD often experience difficulties in psychosocial and occupational functioning, as well as cognitive impairment, and they are characterized by a reduced quality of life [4, 5]. Disfunction in psychosocial functioning have been demonstrated in 30–60% of adults with BD [6] and in 10–15% of patients with BD in remission [7]. Functioning impairments affect various spheres of the lives of patients with BD, such as work, communication, family relationships, recreation, as well as other social activities [8, 9]. Some data indicate that even with complete clinical remission, in 30–50% of patients with BD the premorbid level of psychosocial functioning is not restored, which leads to a reduced ability to assume a normal workload [10]. According to MacQueen et al., 30–60% of patients with BD experience social and occupational difficulties [11]. There is some indication that social adaptation proceeds better in patients with a higher level of education who enjoy the presence of a family or are in a civil marriage, with a shorter duration of the disease [12]. Mood fluctuations and shattered self-esteem are present in patients with BD in remission [13]. Residual symptoms and impairments in social cognition negatively affect the psychosocial functioning of patients with BD [5]. Clinically euthymic patients with BD continue to show impaired Quality of Life (QOL) [14], which is attributed to residual depressive and cognitive symptoms [15].
The concept of burden of illness (BOI) is used to assess the impact of health-related problems at the individual and social levels [16]. Researchers distinguish between the epidemiological (encompassing both the years of life lost due to the disease as well as the morbidity) and economic (direct and indirect costs as well as health care resource utilization) burden of the illness [17]. A population-based approach to estimating the burden of the disease using measurements such as Quality of Life Adjusted Years (QALY) [18] and Disability Adjusted Years (DALY) [19] is widely used; however, these measurements are not fully applicable to an individual patient’s experience of the full burden of illness [20]. In this regard, the development of an individualized means of assessment of the burden of illness appears relevant.
The concept of the Individual Burden of Illness Index (IBI index) was first proposed by Ishak et al. [20]. The IBI index was specifically designed and validated for major depressive disorder [20], and its constituent parts have undergone initial validity testing and are recommended for assessing the functional remission status of patients with recurrent depression in Russia [21]. The use of the index in patients with BD would allow one to objectify their functional state on the basis of an integral assessment.
The aim of this study was to approve and validate the IBI index for the integral assessment of disease burden in patients with BD in remission (IBI-BD index).
METHODS
Study design
A cross-sectional study was conducted.
Setting
The study was conducted in the outpatient psychiatric services of Psychiatric Hospital No. 1 named after P.P. Kaschenko in Saint Petersburg, Russia. The patients in the study were recruited from April through October 2020. The patients were examined during the follow-up period in a community treatment setting to prevent disease relapse.
Participants
General information
Although only a small number (5%) of patients in the original study by Ishak et al. [20] were in remission, since there are no other studies concerned with validation of the IBI index in patients with BD, in the current research, the authors chose to concentrate on patients with BD in remission. This decision was dictated by the widespread interest in evaluating the functioning of individuals with BD during remission, as well as the impact of lingering residual mood symptoms on their overall functioning [22].
Eligibility criteria
The inclusion criteria were
- compliance of the patient’s mental state with BD remission according to International Classification of Diseases-10 (ICD-10);
- symptom severity less than 7 points on the Hamilton Depression Rating Scale (HDRS) [23];
- symptom severity less than 12 points on the Young Mania Rating Scale (YMRS) [24].
The non-inclusion criteria were
- the presence of a comorbid psychiatric disorder;
- the presence of an actual somatic disease or exacerbation of a chronic disease.
The exclusion criteria were
- patients’ refusal to participate in the study at any stage;
- identification of signs of a comorbid mental and/or substance use disorder during the clinical interview.
The criteria for remission assessment were based on the clinical guidelines for the treatment of BD approved in Russia1.
Selection of participants in groups
Eighty-five patients with BD type I (75%; n=64) and BD type II (25%; n=21) in remission were examined.
Variables
The outcome is the calculation of an IBI-BD index, which can take any positive or negative value (for more information see Table S1 in the Supplementary).
Data sources/measurement
General information
The invitation to participate in the study was extended to patients with a confirmed diagnosis of BD by the psychiatrists who provided supportive treatment in the community. After securing patient consent to participate in the study, a face-to-face meeting between the patient and the psychiatrist-researcher (who was not involved in the treatment of the patient) was arranged at the outpatient psychiatric center. Participation in the study involved a one-time clinical interview with a psychiatrist-researcher with a structured interview and the use of clinical scales. The structured interview included the collection of socio-demographic characteristics (sex, age), as well as age of onset, and duration of the disease. During the clinical interview, a psychiatrist-researcher had to confirm that the patient met the criteria for a diagnosis of BD. Since there is no differentiation between BD types I and II in ICD-10 but their diagnosis is determined by an important stage of treatment planning according to clinical recommendations in Russia1, the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM, 5th Edition) criteria were used to confirm the type of BD. The study was conducted in Russian.
Individual Burden of Illness Index adaptation
The process of IBI-BD index adaptation preceded the patient recruitment phase of the study and the validation of the index. Although other methodologies were used in Ishak’s original study [20] (Quick Inventory of Depressive Symptomatology-Self Report, QIDS-SR, for depressive symptom severity; the Work and Social Adjustment Scale, WSAS, for functioning; and the Quality of Life Enjoyment and Satisfaction Questionnaire — Short Form, Q-LES-Q, for quality of life), the authors of the present study elected to replace them, because the methodologies from the original study, to the best of the authors’ knowledge, had not been previously translated and validated for use in the Russian-speaking population. The methodologies chosen by the authors of this study on the contrary are widely used in the practice of psychiatry in Russia [25–29], not least because they are covered by clinical guidelines1, meaning that their use in clinical practice will not require additional time resources.
The World Health Organization (WHO) describes QOL as how people perceive their existence in light of the cultural and value norms surrounding them, considering their aspirations, expectations, standards, and worries2. This concept encompasses various aspects, such as physical well-being, emotional state, individual beliefs, personal autonomy, social connections, and the living conditions they experience [30]. The WHO’s Quality of Life Questionnaire, (WHOQOL)2 was used to assess the QOL. The WHOQOL-100 is an extensive version of the WHOQOL assessment tool designed to provide a detailed and comprehensive understanding of an individual’s QOL within their specific cultural, social, and personal contexts. The WHOQOL-100 consists of 100 questions. Specifically, the scale generates six domain scores, 24 specific facet scores, and a single overall score that assesses general health and quality of life. The six domain scores capture an individual’s self-reported quality of life across six key areas: physical, psychological, level of independence, social relationships, environment, and spirituality. Each domain and facet scores are scaled in a positive direction, with higher scores indicating a higher quality of life.
The severity of affective symptoms was assessed using the HDRS and the YMRS as reflected in the clinical guidelines for the diagnosis and treatment of BD1. The HDRS is a widely used clinician-administered scale for assessing the severity of depressive symptoms in individuals with major depressive disorder or other mood disorders. The HDRS consists of 21 items that evaluate various aspects of depression, such as mood, cognitive symptoms, somatic symptoms, and suicidal ideation. The scale ranges from 0 to 53, with higher scores indicating more severe depressive symptoms. The YMRS is an 11-item clinician-rated scale specifically designed to assess the severity of manic or hypomanic symptoms in individuals with BD or other mood disorders. The YMRS evaluates various aspects of mania, such as mood elevation, irritability, and behavioral disturbances. The scale ranges from 0 to 60, with higher scores indicating more severe manic symptoms. The HDRS and YMRS are useful tools for monitoring the progress of patients in treatment and evaluating the efficacy of interventions.
To assess social functioning, the Personal and Social Performance Scale (PSP) was used [31]. PSP is an instrument designed to assess the functional outcomes and social adjustment of individuals with severe mental disorders assessed over the past 7 days in 4 main areas of social functioning: socially useful activities, relationships with relatives and other social relationships, self-care, and disturbing and aggressive behavior. Scores are given on a scale from 1 to 100, divided into 10 equal intervals, where each interval corresponds to a certain degree of difficulty in social functioning. Higher scores indicate higher levels of functioning.
Bias
No factors were used to stratify the sample. Remission boundaries were chosen according to the recommended cut-off points1. Since it was assumed that any level of quality of life and social functioning could be in remission, no cut-off points or groupings were used for these characteristics.
Statistical analysis
Study size
Since no similar studies have been conducted for patients with BD, it was not possible to perform the target sample size calculations. Therefore, we opted for empirical rules of thumb to determine the sample size. We defined a threshold of at least 80 observations, which is double the minimum sample size value [32].
Statistical methods
Statistical analysis was performed using the R v.3.6.1. (R Core Team, 2020). The mathematical and statistical analysis was performed by a bio-medical statistician who was not involved in data collection and only had access to numerical measures. Absolute values and fractions of the whole, n (%), were used to describe categorical variables. Variables with continuous distribution were described by mean (Mean) and standard deviation (SD); discrete variables and ordered data — by median, 1–3 quartiles (Md [Q1; Q3]). The normality of sample distribution was evaluated using the Shapiro–Wilk test and considered when choosing a method. Data were normally distributed, except where specified otherwise. We used Chi Square (χ2) tests for categorical variables. The Mann–Whitney test was used to compare quantitative data. Correction for multiple hypothesis testing was performed using the Benjamini–Hochberg correction (false discovery rate).
The Kaiser–Meyer–Olkin criterion and Bartlett’s sphericity criterion were used to measure sampling adequacy. The index was calculated using principal component analysis (PCA). The aim of PCA is to extract important information from the observed variables and represent it as a set of new orthogonal variables called principal components. In contrast to describing the variables separately, the data reduction technique provides a composite description of the observed pattern of values. Since scale scores by their nature belong to the ordered scale [33], a nonlinear version of PCA [34], which is implemented in the Gifi package3, was used. A linear transformation of the original data was performed to extract the two components. The resulting eigenvalue was used to estimate the explained variance, and loadings showed the contribution of each variable to the extracted components. The validity of component extraction was also verified. For this purpose, a null distribution was generated from the original data by sequentially shuffling the data in each column independently (the so-called permutation of a single variable strategy) [35]. A total of 999 iterations were performed (separately for each variable), and the starting value of the random number generator (set. seed) was 4,321. The observed eigenvalue was compared with the obtained null distribution. In this case, the one-tailed hypothesis about the superiority of the observed value over the center of the null distribution is tested [36]. Usually, the p-value is calculated as (q+1)/(i+1), where “q” is the number of values from the null distribution that are greater than or equal to the observed value, and “i” is the number of iterations performed [37]. Since the one-tailed backward hypothesis can be tested, a two-tailed p-value was calculated to ensure a more reliable result. Only those components that were significantly greater than the null distribution were selected for further analysis. “1” was added to the numerator and denominator, because the p-value during the Monte Carlo permutation cannot be equal to zero [38].
Linear regression was used for simple conversion of initial scores into the final index. It was tested for the conformity of the residuals to the normal distribution (Shapiro–Wilk test) and homoscedasticity (Breush–Pagan test) [39]. To assess the influence of the BD type and clinical and functional characteristics, separate logistic regressions (proportional odds logistic regressions) without interaction between independent variables were used. The rationale for using this model instead of the classical linear model is based on two considerations. Since the IBI-BD for these patients is calculated for the first time, the assumptions of normality of distribution and homoscedasticity are both strong and optional. The robustness of the model is due to the use of only guaranteed ordering information, which is invariant to any monotonic transformation [40]. The model with predictors was compared with the model without predictors using the log-likelihood test. Regression coefficients and their standard error (b(se)) are presented as the logarithm of the odds ratio [log(odd)]. Null hypotheses were rejected at p <0.05, with additional attention paid to results where null hypotheses were rejected at p <0.005.4
Ethical approval
Patients were included in the study after signing an informed voluntary consent form. The study protocol was approved by the Ethical Committee of Saint Petersburg State University (Protocol No. 02-195; March 16, 2020).
RESULTS
Participants
Eighty-five patients with BD type I (75%; n=64) and BD type II (25%; n=21) in remission were examined. The study sample consisted of 29 males and 56 females, aged from 18 to 45 (mean age 36.6±5.7 years).
Descriptive data
The main sociodemographic characteristics of the sample and the results of the scale score are summarized in Table 1. These data are in fact descriptive statistics of the IBI-BD index. Patients’ age, age of onset, disease duration, sex distribution, and mean values of the scale scores did not differ between the comparison groups; so, further analysis was performed on the entire sample without taking into account the BD type. The mean value of social functioning on the PSP scale in the sample corresponded to the presence of mild difficulties in one or more of the areas of social functioning.
Table 1. Socio-demographic and clinical and scale characteristics of patients with BD types I and II
|
Parameter |
BD type I (n=64) |
BD type II (n=21) |
Statistical test |
|
Age (Mean [SD]) |
37.3 [6.5] |
34.5 [6.8] |
t=842, df=84, p=0.195 |
|
Age of the BD onset (Mean [SD]) |
27.4 [4.6] |
27.1 [5.5] |
t=692, df=84, p=0.982 |
|
Duration of the disorder (Mean [SD]) |
9.9 [5.0] |
7.3 [4.3] |
t=885.5, df=84, p=0.171 |
|
Sex, n (%) |
|||
|
Male |
24 (37.5%) |
5 (23.8%) |
χ²=0.8, df=1, p=0.377 |
|
Female |
40 (62.5%) |
16 (76.2%) |
χ²=0.8, df=1, p=0.377 |
|
HDRS (Md [Q1; Q3]) |
3.0 [2.0; 4.0] |
2.0 [2.0; 4.0] |
U=776.5, p=0.394 |
|
YMRS (Md [Q1; Q3]) |
2.0 [1.75; 3.0] |
2.0 [2.0; 3.0] |
U=670.5, p=0.992 |
|
WHOQOL (Md [Q1; Q3]) |
63.5 [59.4; 68.4] |
66.8 [60.3; 69.8] |
U=560.5, p=0.394 |
|
PSP (Md [Q1; Q3]) |
75.5 [73.0; 79.0] |
79.0 [75.0; 81.0] |
U=479.0, p=0.171 |
Note: BD — bipolar disorder; HDRS — Hamilton Depression Rating Scale; YMRS — Young Mania Rating Scale; WHOQOL — World Health Organization’s Quality of Life Questionnaire; PSP — Personal and Social Performance Scale. Benjamini–Hochberg multiple comparison correction was used in the calculations.
Main results
Factors contributing to the burden of illness in bipolar disorder
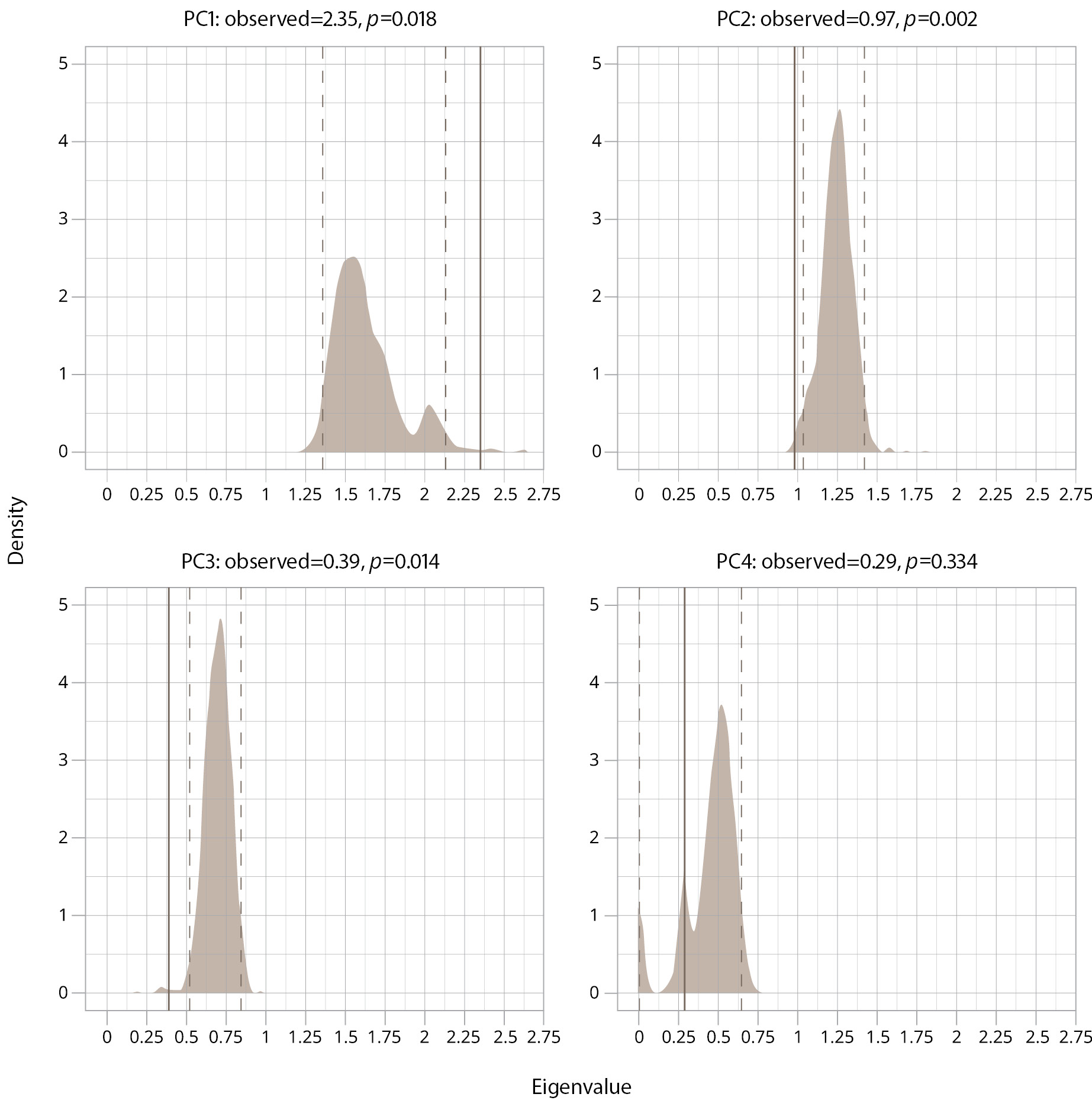
At baseline, the Kaiser–Meyer–Olkin criterion: overall MSA=0.66; YMRS — 0.61; HDRS — 0.63; WHOQOL — 0.77; PSP — 0.64 (all values exceed the mediocre level); and Bartlett’s sphericity criterion — 98.67 (6), p <0.001. The mean scores of the HDRS, YMRS, PSP, and WHOQOL-100 were included in the analysis of the factors contributing to the burden of illness in BD in remission. A graphical representation of the eigenvalue comparison with the null distribution by the bootstrap method is presented in Figure 1. According to the data obtained during the enumeration, the eigenvalue of the principal component 1 (PC1) exceeds the upper limit of the 95% confidence interval of the null distribution. The observed values for the other components either do not exceed the boundaries of the null distribution or are on its lower boundary. Since the values exceeding the “noise data” are the ones of interest, only PC1 values were used in further analysis.
Figure 1. A graphical representation of the eigenvalue comparison with the null distribution by the bootstrap method in the sample. Note: PC — principal component; p — p-value; shade area — null distribution; solid line — observed eigenvalue; dashed line — 95% confidence interval under null distribution.
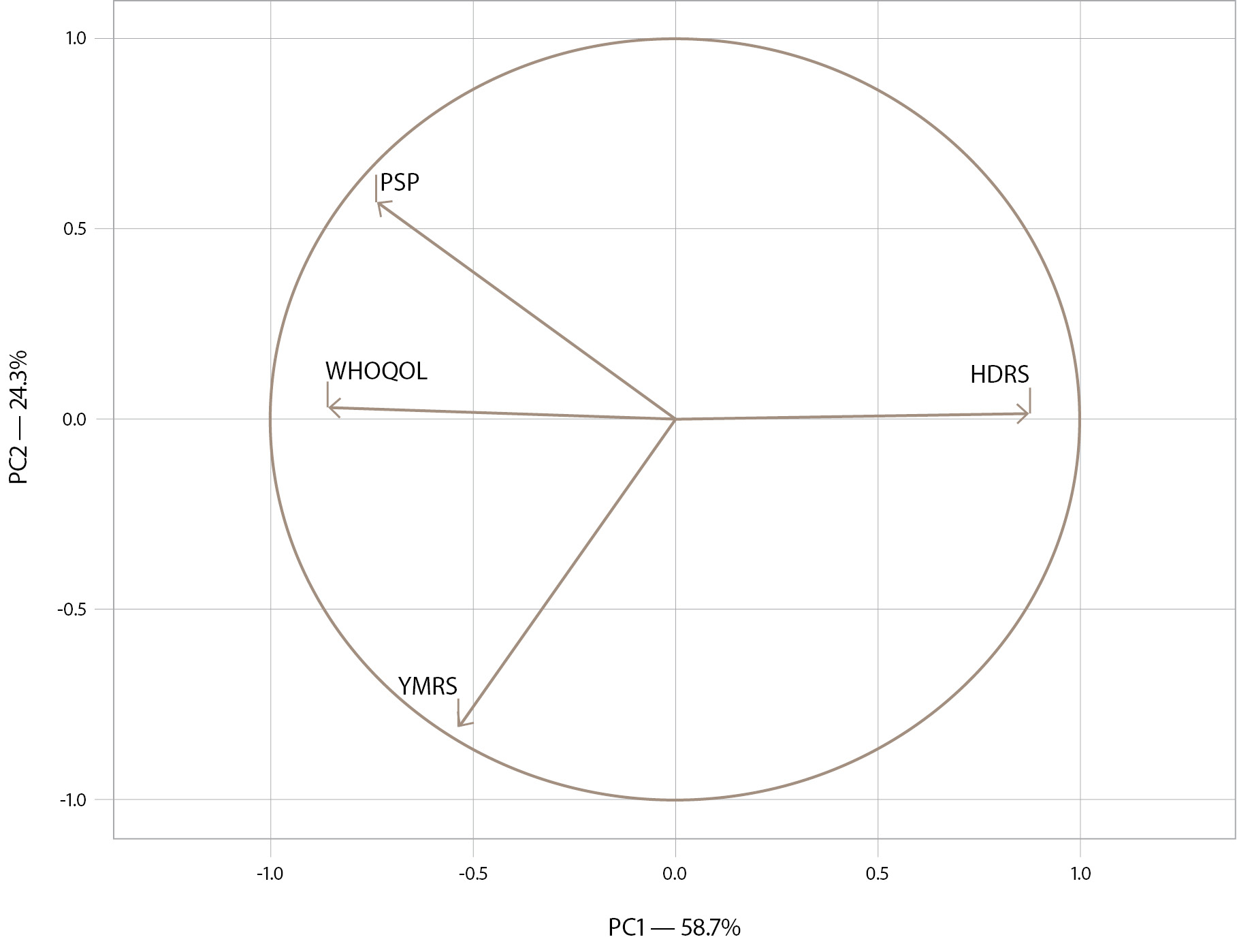
Principal component analysis in accordance with the adjusted variables is presented in Figure 2. The loading plot in Figure 2 shows that the burden of illness in remission is directly related to the severity of residual depressive symptoms (as the HDRS score increases, social functioning and quality of life decrease). Conversely, the presence of residual mania symptoms (YMRS score) is associated with a lower burden and higher level of social functioning and quality of life. The burden index explains 58.7% of the variance in the data (X-axis).
Figure 2. The loading plot of the burden of illness in patients with BD in remission. Note: HDRS — Hamilton Depression Rating Scale; YMRS — Young Mania Rating Scale; WHOQOL — World Health Organization’s Quality of Life Questionnaire; PSP — Personal and Social Performance Scale; PC – principal component.
To calculate the IBI-BD index directly, we can use the equation from Table 2. The table shows the linear regression coefficients by which the scale scores should be multiplied. The obtained values are added up, and a constant is added. Thus, the equation for calculating the IBI-BD index looks as follows:
IBI-BD index = 6.64 + (-0.14 × YMRS) + (0.27 × HDRS) + (-0.04 × QOL) + (-0.06 × PSP)
Table 2. Equation for calculating the IBI index for patients with BD in remission
|
Variable |
Statistic |
p-value |
|
(Intercept) |
6.64 (0.69) |
p <0.001 |
|
YMRS |
-0.14 (0.03) |
p <0.001 |
|
HDRS |
0.27 (0.03) |
p <0.001 |
|
WHOQOL |
-0.04 (0.00) |
p <0.001 |
|
PSP |
-0.06 (0.01) |
p <0.001 |
|
Shapiro–Wilk test |
W=0.98 |
p=0.193 |
|
Breusch–Pagan test |
χ²=7.8 (df=4) |
p=0.101 |
|
Fisher test |
F=207.5 (4; 80) |
p <0.001 |
|
The coefficient of determination (adj.) |
R²=0.91 |
- |
Note: IBI — Individual Burden of Illness; BD — bipolar disorder; HDRS — Hamilton Depression Rating Scale; YMRS — Young Mania Rating Scale; WHOQOL — World Health Organization’s Quality of Life Questionnaire; PSP — Personal and Social Performance Scale.
This model satisfies the theoretical premises of linear regression (according to insignificant Shapiro–Wilk and Breusch–Pagan tests). Also, the linear model relates well the initial values with the final indicator (coefficient of determination >0.9). This equation allows for a quick calculation of the IBI-BD index value in case of lack of access to baseline data or inability to perform PCA. Because the IBI-BD index is based on a z-score, it is easy to calculate a patient’s burden of illness relative to other patients with BD in remission. An IBI-BD index with a negative value indicates that the patient has a lower disease burden compared to the average patient seeking treatment, whereas an index with a positive value indicates that the patient’s disease burden is higher.
The assessment of the influence of the evaluated characteristics on the IBI-BD index is presented in Table 3. The search for the dependence of the IBI-BD index on the main clinical and demographic characteristics was performed using proportional odd logistic regression, taking into account the diagnostic group. According to the obtained data, the sex and age of disease onset could not be associated with IBI-BD, as no superiority of the analyzed models over the models without predictors was revealed. The other indicators were statistically significantly associated with IBI-BD. In each model, the regression coefficient describing the intergroup difference is not different from zero (p >0.05). This implies that no significant intergroup difference in IBI between BD type I and BD type II diagnoses can be inferred. Patients’ age [log(odd)=0.11(0.03)], as well as disease duration [log(odd)=0.18(0.04)], was directly related to the IBI-BD index value. The directions of association of the last four indicators do not differ from those in linear regression modeling (see Table 2).
Table 3. Assessment of the impact of the estimated characteristics on the IBI-BD index
|
Variable |
Model test |
BD type I vs BD type II; log(odd)(se), p |
Key — log(odd)(se), p |
|
Sex |
χ²=4.9 (df=72), p=0.084 |
- |
- |
|
Age |
χ²=18.2 (df=72), p <0.001 |
-0.77 (0.48), p=0.109 |
0.11 (0.03), p <0.001 |
|
Age of BD onset |
χ²=5.3 (df=72), p=0.079 |
- |
- |
|
Duration of the disorder |
χ²=22.5 (df=72), p <0.001 |
-0.49 (0.49), p=0.316 |
0.18 (0.04), p <0.001 |
|
YMRS |
χ²=31.0 (df=72), p <0.001 |
-0.90 (0.47), p=0.057 |
-0.88 (0.18), p <0.001 |
|
PSP |
χ²=63.0 (df=72), p <0.001 |
-0.23 (0.46), p=0.619 |
-0.39 (0.06), p <0.001 |
|
HDRS |
χ²=94.6 (df=72), p <0.001 |
-0.67 (0.48), p=0.162 |
1.49 (0.18), p <0.001 |
|
WHOQOL |
χ²=51.0 (df=72), p <0.001 |
-0.65 (0.48), p=0.174 |
-0.19 (0.03), p <0.001 |
Note: IBI — Individual Burden of Illness; BD — bipolar disorder; HDRS — Hamilton Depression Rating Scale; YMRS — Young Mania Rating Scale; WHOQOL — World Health Organization’s Quality of Life Questionnaire; PSP — Personal and Social Performance Scale; Benjamini–Hochberg multiple comparison correction was used in the calculations.
DISCUSSION
Key results
The present study focuses on the approval and validation of the IBI index in patients with BD in remission, which was previously developed and validated for major depressive disorder. To the best of the authors’ knowledge, this is the first time such work has been done. The IBI-BD index is a simple multidimensional metric based on patient-reported outcomes used to describe the complexity of affective disorder as an illness, including the burden it imposes on the individual by incorporating symptoms’ severity, functioning, and QOL impairments [41]. The research yielded important findings for clinical practice, most notably the fact that residual depressive and manic symptoms differentially affect functioning and quality of life in individuals with BD in remission. Moreover, the BD type does not make an additional contribution to this state of affairs. And the validated IBI-BD index could be applied in clinical practice for a more personalized assessment of the BD in remission individual disease burden.
Strengths and limitations
A key strength of this study is that to the best of our knowledge it is the first study to address the individual burden of illness for patients with BD in remission. Another advantage is that the study included patients with both types of BD. We recognize, however, that the study has a number of potential limitations. The cross-sectional design of the study is among the limitations. The authors are aware that the sample size is rather small. However, the analysis showed statistically reliable results and the ability to draw conclusions even with such a sample. The research was conducted on patients in remission, which means the results cannot be directly applied to all individuals with BD.
The set of scales used in the present study differs from the original study [20], but the authors believe this discrepancy probably did not compromise the integrity of the findings. The instruments for calculating the IBI replacement are justified for the following theoretical reasons: undoubtedly, the concept of the IBI itself is theoretical and the options for its computation may not be limited to baseline scales or baseline diseases. The essential point is to link disease symptoms, quality of life, and social functioning into a consolidated assessment system that is not reduced to a one-dimensional comparison of individual parameters. Scales and questionnaires in their original form do not have the property of equidistance (i.e., the difference in scores does not indicate the true distance between 2 dimensions), but they do have the property of ranking. The ranking property is the unobserved metric of “depression”, “quality of life”, etc. When performing data reduction, we discard the original units of measurement and reach for some normalized values. Assuming the scales measure the same thing, we should obtain roughly comparable results (at the least, the same if the units differed by a constant, e.g., instead of kg–pounds, degrees Kelvin–Celsius, etc.). For scales, the number of categories claimed and self/external scoring can potentially influence the result. In our case, self-questionnaires were replaced by clinical scales recommended by clinical guideline5, potentially affecting the adaptation results. However, the high statistical significance of our results demonstrates the feasibility of this approach.
We also did not take into account the influence of the pharmacotherapy received by the patients due to the considerable individual differences between the patients. Other clinical variables that potentially affect the burden of BD (presence of comorbid disorders, number of episodes, number of hospitalizations, etc.) were not assessed in relation to the IBI-BD index, because they were not included in the original study. Assessing the influence of these variables on the IBI-BD index could be one of the future directions of research. It is known that cognition, when objectively measured, is severely impaired in BD [42] and has also been associated with occupational outcomes. This suggests that cognitive functioning may also potentially contribute to the individual disease burden. However, since this aspect was not considered in the initial index, it was also disregarded in our study.
Interpretation
According to Ishak et al. [20], the concept of individual burden of illness represents the overall impact of a disease, which includes the suffering caused by symptom intensity, frequency, and duration; limitations in occupational, social, and leisure activities; and the patient’s overall satisfaction with health, work, social life, and recreational pursuits. To quantify this concept, Ishak et al. [20] developed the IBI index through a principal component analysis of patient-reported data on symptom severity, functioning, and quality of life, using it as a mathematical abstraction based on other psychometric scales.
The main goal of a doctor in clinical practice is to minimize symptoms, improve social functioning, and select a treatment adequate to the patient’s condition. The relevance of this work is that the burden of disease at the individual level was calculated in patients with BD in remission. Given the new data on the frequency of residual [41] and subthreshold [22, 43, 44] symptoms in patients with BD in remission, the very notion of the limits of remission in BD is widely debated in the scientific literature. A growing body of evidence indicates that during remission, patients with BD often present subsyndromal mood symptoms, which are associated with poor psychosocial functioning, cognitive impairment, and reduced quality of life [45–48]. The validated IBI-BD index helps to assess the burden of negative factors on remission.
When compared with the original study of the application of the IBI index for patients with major depressive disorder [20], a limited explanation of data variance can be observed in our study. Possible reasons for this are as follows: mixing external ratings with self-reported questionnaires, and extreme heterogeneity of the parameters assessed. Since only the PC1 is greater than the zero distribution, its use for the IBI-BD index is consistent with an earlier study [20].
Depressive symptoms, including subsyndromal ones, are responsible for most of the burden that is associated with BD in terms of functioning, QOL, economic loss, and suicide [3]. The previous study suggested that personal recovery among patients with BD is affected by stigma, level of functioning, residual depressive symptoms, and employment status [49]. Functional impairment is an important driver of disability in patients with BD and can persist even when symptomatic remission has been achieved [50]. In our study, it was found that social functioning and quality of life decreased as the total score on the HDRS increased. At the same time, the presence of residual symptoms of hypomania (e.g., increased daytime activity and sexual interest) is subjectively evaluated by patients as positive phenomena, and it is also considered by patients as desirable and contrasted with residual depressive symptoms. Our data support the need for management of subsyndromal depressive symptoms in patients with BD even in the inter-episodic period [51]. The results of our study further contribute to the understanding of how residual affective symptomatology affects the functioning of patients with BD in remission, and it demonstrates the need to develop more targeted guidelines for the assessment and treatment of residual (subthreshold) symptoms.
When working with patients with BD, we need to bear in mind that BD is a complex psychiatric condition with a high heterogeneity in its manifestation, and that the BD II subtype may lead to similar health (and social) consequences as the BD I subtype [52]. In our study, this was confirmed, as patients with both types of BD showed no differences in functioning and no intergroup differences in the IBI-BD index.
The authors of the study consider the clinical significance of the validated IBI-BD index to reside in providing physicians with an additional technique for assessing patients’ condition and grading its severity even when the remission criteria are formally met. Since the HDRS and YMRS scales are already included in the recommended scales for the assessment of patients with BD in clinical practice, the additional application of easy-to-use methods (WHOQOL, PSP) in the opinion of the researchers will not significantly increase the clinician’s workload. Introducing the use of the IBI-BD index into clinical practice will allow additional interventions to be justified from the perspective of the patient’s personal burden of illness and will allow interventions to be more personalized in the context of the lack of algorithms for the treatment of residual affective symptomatology in the remission of BD.
The use of the IBI index is not limited to assessing burden of illness and has already been tested in assessing the effectiveness of therapy [53] and predicting relapse in major depressive disorder [54]. Further work for researchers after the approval and validation of the index for patients with BD is seen in expanding opportunities for the scientific and practical use of the index, including the introduction of methods for its calculation in routine clinical practice. The practical application of the presented study is seen in the utilization of the IBI-BD index in clinical practice in order to objectify the functional status of patients with BD in remission.
Generalizability
The results of this study can be applied to comparable patients with BD for the following reasons: first, the validated tools were used to assess residual symptoms, quality of life, and social functioning. Second, we proposed a simple linear equation linking the disease burden index to its components; so, it can be used if methods are available. Third, the methodology for obtaining the IBI-BD was described, making it possible to obtain a similar index on a different set of techniques independently. Despite the differences between the scales and questionnaires, they measure the same latent construct (different for each method); so, there should be no significant differences between the main components in the case of alternative IBI calculation. Fourth, there were no artificial conditions for the study — patients of both sexes with differences in age and disease history participated; that is, the sample was a cross-section of real patients that any physician or researcher may encounter.
CONCLUSION
To the best of authors’ knowledge, this is the first study introducing and validating a composite calculation of the Individual Burden of Illness index in BD in remission. We have demonstrated by statistical means that it is possible to successfully approve and validate the previously calculated IBI index in major depressive disorder for patients with BD in remission. The proposed index assesses both the severity of symptoms and the functioning and QOL in patients with BD, resulting in a single weighted composite score that adequately reflects the disease burden. The study has shown that residual affective symptoms have a differing impact on the functioning of patients with BD in remission, reflecting the need for timely assessment and targeted therapy of these symptoms in such patients. It was found that social functioning and quality of life decrease in the presence of residual depressive symptoms, while residual symptoms of hypomania have the opposite effect. The results obtained may help to more objectively assess the functional status of patients with BD in remission using a statistical model.